Introduction While the approved dose of brentuximab in T cell lymphomas has been every 3 weeks on a 1.8 mg/kg schedule, earlier studies exploring weekly dosing showed that a dose of 1.2 mg/kg on a weekly dosing (every 3 out of 4 weeks) in pts with Hodgkin's lymphoma and hematologic malignancies may improve cancer response rates while still having manageable side effects3. We explored the weekly dosing schedule in 37 pts (pts) with mycosis fungoides/Sezary syndrome (MF/SS) and aggressive T cell lymphomas and compared to our experience with every 3 week dosing in 36 pts to evaluate tolerability and efficacy of the weekly schedule..
Methods We reviewed charts of 67 pts, 36 received dosing q 3 weeks and 37 received a dose weekly for 3 consecutive weeks on a 4-week schedule. Pts included MF/SS (n=35), gamma delta T cell lymphoma (n=2), anaplastic large cell lymphoma (n=12), Peripheral T cell Lymphoma (n=10), angioimmunoblastic T cell lymphoma (n=4), adult T cell leukemia (n=2), and NK-T cell lymphoma. Pts were treated with brentuximab vedotin at a dose of either 1.8 mg/kg every 3 weeks or 1.2 mg/kg weekly x 3 every 4 weeks. CD30 expression was scored by the pathologist in tumor biopsies as high (<50%), low (5-10%), or intermediate (>10%-49%). Toxicity data was recorded from the medical records and data analyzed descriptively.
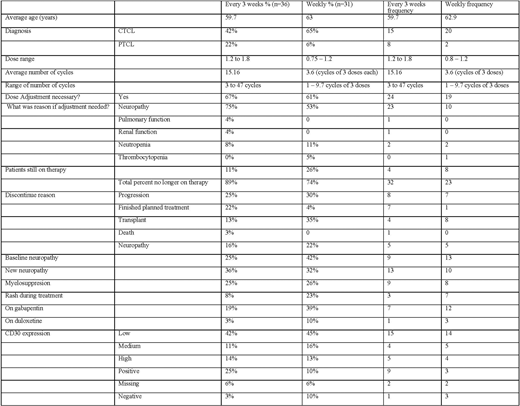
Results Of 67 pts in this study, the average age was 61. Doses were 1.8 mg/kg for the q 3 week schedule and 0.75 to 1.2 mg/kg for the weekly x 3 schedule. Cycles were 3-47 for q 3 weeks and 1-9.7 for weekly dosing. CD30 expression was high in 13% of pts, low in 43%, and absent in 6% with equal distribution between the weekly and q 3 week cohorts, as shown in Table 1. Dose adjustments were made in 67% of q 3 week and 61% of weekly pts for neurotoxicity (n=28), with a higher incidence in the q 3 week pts compared to those with weekly dosing (75% vs 53%, p=0.01) . Discontinuation for progression (25% vs 30%) was similar for both groups. In the weekly group, 8 pts had a stem cell transplant, including allogeneic transplantation in 3.
Conclusion In the Phase II registration trial of brentuximab vedotin 1.8 mg/kg q3 weeks, 41% of pts had neuropathy (severe in 12%). 1 Forty two percent of discontinuations were for neuropathy. In our weekly schedule, incidence of neuropathy was lower and led to fewer treatment discontinuations. Our retrospective data shows that Brentuximab vedotin is well tolerated on a weekly dosing schedule and has activity in pts with MF/SS and aggressive T cell lymphomas. As in prior studies, responses were seen with low CD30 expression4, 5. Prospective clinical trials with a self-reported neurotoxicity scale and quality of life instruments should be performed address the impact of more frequent, lower doses of brentuximab vedotin on patient outcomes.
1 Pro B, Advani R, Brice P, Bartlett NL, Rosenblatt JD, Illidge T et al.J Clin Oncol 2012; 30(18): 2190-2196. doi: 10.1200/JCO.2011.38.0402
2 Prince HM, Kim YH, Horwitz SM, Dummer R, Scarisbrick J, Quaglino P et al.Lancet 2017; 390(10094): 555-566. doi: 10.1016/S0140-6736(17)31266-7
3 Fanale MA, Forero-Torres A, Rosenblatt JD, Advani RH, Franklin AR, Kennedy DA et al.Clin Cancer Res 2012; 18(1): 248-255. doi: 10.1158/1078-0432.CCR-11-1425
4 Duvic M, Tetzlaff MT, Gangar P, Clos AL, Sui D, Talpur R. J Clin Oncol 2015; 33(32): 3759-3765. doi: 10.1200/JCO.2014.60.3787
5 Kim YH, Tavallaee M, Sundram U, Salva KA, Wood GS, Li S et al.J Clin Oncol 2015; 33(32): 3750-3758. doi: 10.1200/JCO.2014.60.3969
Huntington:Pharmacyclics: Honoraria; DTRM: Research Funding; Genentech: Consultancy; Novartis: Consultancy; Celgene: Consultancy, Research Funding; TG Therapeutics: Research Funding; Bayer: Consultancy, Honoraria; AbbVie: Consultancy; Astrazeneca: Honoraria. Xu:Seattle Genetics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal